We cover endometriosis symptoms and treatment with the mechanism-level science most health articles skip — the retrograde menstruation debate, the immune dysfunction hypothesis, the oestrogen-inflammation feedback loop, the Indian diet interventions with the strongest evidence, the Ayurvedic perspective, and the clear explanation of what each treatment actually does and why. Because understanding your condition is the beginning of managing it.
What Endometriosis Actually Is — Beyond the Textbook Definition
The standard definition — “tissue similar to the uterine lining grows outside the uterus” — is accurate but incomplete. It misses the most important clinical truth about endometriosis: it is not simply a gynaecological problem. It is a systemic inflammatory, immune-mediated, oestrogen-dependent disease that affects the entire body — not just the pelvis.
Endometriotic lesions behave like the endometrium in one critical way: they respond to the hormonal cycle. Each month, these lesions receive the same oestrogen signals that cause the uterine lining to proliferate, and the same progesterone-withdrawal signals that trigger shedding. But unlike menstrual blood, which exits the body, the blood and inflammatory mediators from lesions have nowhere to go. They cause local inflammation, scar tissue (adhesions), and immune system activation — cycle after cycle — for years before a diagnosis is typically reached.
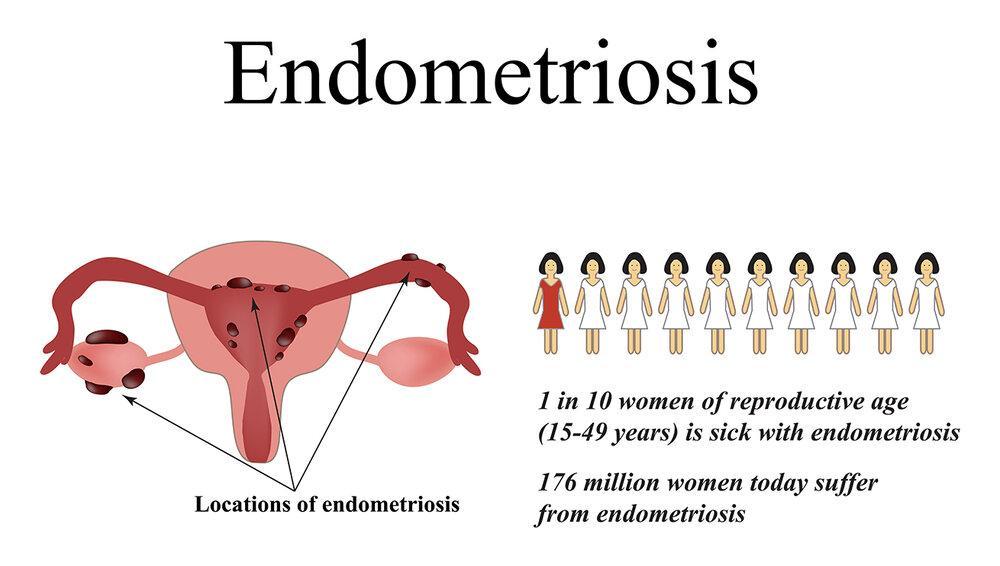
Peritoneal (surface) endometriosis: Lesions on the pelvic peritoneum (the membrane lining the pelvic cavity). Often the most common and may be less immediately symptomatic, but drives the systemic inflammatory burden.
Ovarian endometriosis (endometriomas / “chocolate cysts”): Blood-filled cysts on the ovaries — named for their dark, tar-like content. Can grow to 10cm+. Damages ovarian reserve and is associated with reduced egg quality. Detectable on ultrasound.
Deep infiltrating endometriosis (DIE): Lesions penetrating more than 5mm beneath the peritoneum — typically affecting the uterosacral ligaments, rectovaginal septum, bowel, bladder, or ureters. Causes the most severe pain, especially dyspareunia and bowel symptoms. Requires expert surgical management.
Extra-pelvic endometriosis (rare but documented): Lesions have been found in the diaphragm (causing cyclical shoulder tip pain), the lung (cyclical pneumothorax or haemoptysis), the brain, the surgical scar tissue, and even the nasal mucosa. These diagnoses are frequently missed because physicians do not think “endometriosis” when they see cyclical non-gynaecological symptoms.
What Causes Endometriosis — The Science Beyond “We Don’t Know”
The causes of endometriosis are not fully understood — but this is frequently used as a reason to tell women little. The truth is that research has identified several interacting mechanisms, and understanding them matters because it directly informs treatment choices.
The most cited theory: during menstruation, some endometrial cells flow backward through the fallopian tubes into the pelvic cavity. This retrograde flow is documented in 76–90% of women who menstruate — including women who do not develop endometriosis. This is the critical problem with the retrograde menstruation theory as a sole explanation: most women have retrograde flow but only 10% develop endometriosis. Something else determines who develops the disease.
That “something else” appears to be immune function. In women without endometriosis, natural killer (NK) cells and macrophages in the pelvic cavity recognise and destroy retrograde endometrial cells. In women with endometriosis, this clearance mechanism fails — the immune system does not adequately destroy ectopic endometrial cells, allowing them to implant and proliferate.
⚗️ Retrograde flow in 76–90% of women | NK cell clearance failure as determining factorIncreasing evidence positions endometriosis as fundamentally an immune-inflammatory disease. Women with endometriosis show multiple immune abnormalities: reduced NK cell cytotoxicity in the peritoneal fluid, elevated pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, IL-8) in peritoneal fluid, increased macrophage numbers but impaired function, elevated immunoglobulin levels suggesting autoimmune activity, and regulatory T-cell (Treg) dysfunction that reduces immune tolerance without allowing effective clearance.
The peritoneal fluid of women with endometriosis is literally an inflammatory environment — containing elevated prostaglandins, reactive oxygen species, and cytokines that simultaneously promote lesion survival, drive pain sensitisation, and impair fertility. This is why anti-inflammatory strategies (dietary, pharmacological, and lifestyle) have meaningful impact on endometriosis — they address a core pathological mechanism, not just symptoms.
⚗️ Peritoneal NK cell dysfunction | Elevated IL-1β, IL-6, TNF-α, IL-8 | Autoimmune componentEndometriosis is an oestrogen-dependent disease in two senses: it requires oestrogen to grow, and endometriotic lesions themselves produce oestrogen locally via aromatase enzyme activity — creating a positive feedback loop where the lesion generates its own growth signal. This local oestrogen production means that even in the hypooestrogen environment created by GnRH agonist treatment, lesions may continue producing enough local oestrogen to survive.
The oestrogen-progesterone imbalance adds complexity: endometriotic cells show progesterone resistance — reduced expression of progesterone receptors that normally counteract oestrogen stimulation. This is why progesterone-based treatments (the mainstay of hormonal management) work for many women but not all, and why some lesions continue growing even on combined hormonal contraception.
⚗️ Aromatase-driven local oestrogen production | Progesterone receptor resistance | Self-sustaining growth loopEndometriosis has a clear hereditary component. First-degree relatives (mother, sisters) of women with endometriosis have approximately 7x higher risk of developing the condition. Genome-wide association studies (GWAS) have identified multiple genetic loci associated with endometriosis risk, including variants in genes involved in oestrogen metabolism, immune function, and cell adhesion. However, genetics is not destiny — the condition also requires specific environmental and hormonal triggers to manifest.
For Indian women: family history is one of the strongest risk factors. If your mother or sister has severe painful periods, endometriosis, or unexplained infertility — your own painful periods deserve thorough gynaecological investigation rather than normalisation.
⚗️ 7x risk increase in first-degree relatives | GWAS: oestrogen metabolism + immune gene variantsDioxins and polychlorinated biphenyls (PCBs) — persistent environmental toxins from industrial processes, waste incineration, and certain plastics — act as endocrine disruptors that mimic or amplify oestrogen signalling. Animal studies demonstrated that dioxin exposure at low doses produces endometriosis in primates. Human epidemiological data links higher dioxin body burden to increased endometriosis prevalence and severity. These toxins accumulate in fatty tissue and pass up the food chain — concentrating in animal fats (dairy, meat, fish).
This provides one mechanistic rationale for the observational finding that red meat and high-fat dairy intake are associated with higher endometriosis risk in some cohort studies — beyond just dietary fat content, the bioaccumulation of endocrine-disrupting chemicals in these food sources may be a contributing factor.
⚗️ Dioxin: endocrine disruptor + primate endometriosis model | PCB bioaccumulation in animal fatsEndometriosis Symptoms — The Full Picture Including the Ones That Get Missed
The most important thing to understand about endometriosis symptoms is that pain severity does not correlate with disease stage. A woman with Stage 4 (severe) endometriosis may have minimal pain. A woman with Stage 1 (minimal) endometriosis may be completely debilitated. This is why the disease is so frequently dismissed — women in severe pain are sometimes told “the ultrasound looks normal” or “we can only see a small amount of tissue” without understanding that visible disease burden does not predict pain burden.
How Endometriosis Is Diagnosed — The Honest Guide to a Difficult Process
Endometriosis cannot be definitively diagnosed without surgery. This is the fundamental challenge — and one reason the diagnostic delay is so long. Symptoms, examination, and imaging can raise strong suspicion, but only laparoscopy with histological biopsy provides a confirmed diagnosis. Understanding this pathway helps women navigate the process more effectively.
A detailed menstrual and symptom history is the single most important step in raising endometriosis suspicion. A doctor who takes a thorough history — asking specifically about pain severity, pain timing relative to the cycle, dyspareunia, bowel symptoms at menstruation, family history, and infertility — can identify a high-probability endometriosis profile before any investigation is ordered.
If you are presenting to a doctor, prepare your symptom history in writing. Document: when pain occurs relative to your cycle, pain score on a 0–10 scale, how many days per month pain is present, whether pain is relieved by standard painkillers (NSAIDs), whether you have pain during sex, whether you have bowel or bladder symptoms at menstruation, your family history, and any fertility history. A structured symptom history dramatically increases the likelihood of appropriate investigation.
Transvaginal ultrasound performed by an experienced sonographer is the first-line investigation for suspected endometriosis. It can reliably detect: endometriomas (ovarian endometriotic cysts — appear as homogeneous ground-glass echogenicity on TVUS), some forms of deeply infiltrating endometriosis in the bladder and rectovaginal septum when performed with specific endometriosis protocols (the “soft markers” technique — asking the patient to indicate pain during probe movement, assessing ovarian mobility).
What TVUS cannot detect: peritoneal surface endometriosis (the most common form), small lesions, and early disease. A normal transvaginal ultrasound does NOT exclude endometriosis. Many women are told “the ultrasound is normal” and reassured — this is clinically misleading if the clinical picture suggests endometriosis.
⚗️ TVUS: sensitive for endometriomas, less so for peritoneal disease | Normal US does not exclude endometriosisPelvic MRI with endometriosis-specific protocol (including gel in the rectum and bladder filling) provides the most detailed pre-operative mapping of endometriosis — particularly for deeply infiltrating disease involving the bowel, bladder, and pelvic sidewall. It guides surgical planning and helps identify the need for multi-disciplinary teams (including bowel surgeons) before operating.
MRI is not a screening tool and is not typically first-line unless there is clinical suspicion of deep infiltrating endometriosis. Like TVUS, a normal MRI does not exclude peritoneal endometriosis or early-stage disease.
Diagnostic laparoscopy — a minimally invasive surgical procedure performed under general anaesthesia, in which a camera is inserted into the pelvic cavity through small abdominal incisions — remains the gold standard for endometriosis diagnosis. Direct visualisation of lesions, combined with histological biopsy confirmation, provides definitive diagnosis.
Importantly, the ideal diagnostic laparoscopy is also an excision laparoscopy — performed by a surgeon experienced in endometriosis who can remove lesions at the time of diagnosis rather than simply confirming their presence and closing. “See and treat” laparoscopy produces better symptom outcomes than “diagnose and plan later” approaches. When seeking laparoscopy, ask specifically whether your surgeon will excise (not just ablate/burn) any lesions found, and whether they have specific endometriosis surgical training.
⚗️ Gold standard: laparoscopy + histology | Excision superior to ablation for recurrence ratesTreatment Options — What Each One Does and Who It’s Best For
| Treatment | How It Works | Best For | Key Limitations | Recurrence After Stopping |
|---|---|---|---|---|
| NSAIDs (ibuprofen, naproxen) | COX-2 inhibition reduces prostaglandin-driven inflammation and uterine cramping | Mild–moderate dysmenorrhoea; short-term management | Does not affect lesion growth; GI side effects with regular use | N/A — symptomatic only |
| Combined oral contraceptive pill (COCP) | Suppresses ovulation; reduces menstrual bleeding and lesion stimulation | Mild–moderate symptoms; contraception also required | Does not eradicate lesions; some women do not tolerate oestrogen; not suitable during fertility planning | High — symptoms typically return within months of stopping |
| Progestin-only therapy (pill, injection, hormonal IUD, implant) | Suppresses endometrial growth via progesterone receptor activity; induces decidualisation and atrophy of lesions | Those who cannot tolerate oestrogen; long-term management; menorrhagia control | Progesterone resistance in some lesions; mood effects in some women; irregular bleeding initially | High — disease resumes on discontinuation |
| GnRH agonists (e.g., Leuprolide/Lupron) | Suppress LH/FSH → oestrogen falls to postmenopausal levels → temporary menopause-like state → lesion regression | Severe symptoms; pre-surgical downstaging; diagnostic trial in some cases | Hypoestrogenic side effects (hot flushes, bone loss); expensive; typically limited to 6 months without add-back therapy; does not eradicate disease | High — regrowth begins after oestrogen restoration |
| Laparoscopic excision surgery | Physical removal of lesions, adhesions, and endometriomas — addresses disease burden directly | Moderate–severe disease; fertility desire; failed medical management; diagnostic confirmation | Requires endometriosis specialist surgeon; recurrence at 5 years is 20–40%; multiple surgeries may reduce ovarian reserve (endometrioma surgery specifically) | Moderate — recurrence is lesion regrowth, not surgical failure per se |
| Hysterectomy ± oophorectomy | Removes uterus (± ovaries) — eliminates the hormonal cycle driving lesion stimulation | Severe refractory disease; completed family; adenomyosis co-existing; last resort | Does NOT guarantee cure — peritoneal lesions may remain active if ovaries are retained; immediate surgical menopause if ovaries removed; permanent infertility | Low with bilateral oophorectomy; higher if ovaries retained |
| IVF / ART | Bypasses tubal and natural fertilisation barriers; optimises embryo quality and implantation window | Endometriosis-related infertility not resolved by surgical treatment | Success rates vary by age and endometriosis stage; endometriomas may reduce ovarian response; multiple cycles may be needed | N/A — fertility treatment, not disease management |
Natural and Complementary Management — The Evidence-Based Approach
Natural interventions for endometriosis are most powerful as adjuncts to medical care — not as replacements. The goal is to reduce the systemic inflammatory burden, support oestrogen metabolism, and manage pain through mechanisms that complement rather than conflict with medical treatment. The following are ranked by strength of evidence.
The peritoneal environment in endometriosis is defined by elevated pro-inflammatory cytokines, prostaglandins, and reactive oxygen species. Dietary inflammation is one of the most modifiable drivers of this systemic inflammatory burden. A cohort analysis from the Nurses’ Health Study II found that women consuming the most omega-3 fatty acids had a 22% lower risk of endometriosis diagnosis compared to those consuming the least. Women consuming the most trans fats had a 48% higher risk.
The endometriosis anti-inflammatory dietary framework:
Increase: Omega-3-rich foods (walnuts, flaxseed, chia seeds, mustard oil in cooking); cruciferous vegetables (broccoli, cauliflower, cabbage, kale — their indole-3-carbinol promotes oestrogen detoxification via the 2-hydroxyoestrone pathway); high-fibre foods (lentils, whole grains, vegetables — improve oestrogen elimination through the gut); colourful fruits and vegetables (polyphenols — antioxidant load to counter oxidative stress in peritoneal fluid); turmeric with black pepper daily (curcumin’s NF-kB inhibition directly targets the inflammatory pathway driving endometriosis — see our detailed guide: Health Benefits of Turmeric).
Reduce or avoid: Red and processed meat (arachidonic acid → pro-inflammatory prostaglandins; potential dioxin bioaccumulation); trans fats (directly elevate inflammatory markers); refined sugar (insulin-driven inflammation and IGF-1 elevation may promote lesion growth); excess alcohol (competes with oestrogen for liver metabolism, raising circulating oestrogen); excess caffeine (some studies link to higher oestrogen levels).
Multiple clinical studies and meta-analyses have examined omega-3 fatty acids specifically for endometriosis pain. A 2011 clinical trial found that fish oil supplementation significantly reduced dysmenorrhoea severity and NSAID use in women with endometriosis compared to placebo. A 2018 meta-analysis confirmed that omega-3 supplementation produces significant reduction in menstrual pain — through inhibition of arachidonic acid-derived prostaglandin synthesis (the same mechanism as NSAIDs, but without gastrointestinal side effects).
For vegetarian Indian women: algae-based omega-3 DHA+EPA supplements (400–1,000mg daily) are the most direct alternative to fish oil. Flaxseed oil (1 tbsp daily) provides ALA, which partially converts to EPA and DHA. Walnuts (5–7 daily) provide ALA alongside magnesium. None of these substitutes quite matches fish oil EPA/DHA bioavailability — algae oil is the best vegetarian alternative.
⚗️ Meta-analysis: omega-3 significantly reduces menstrual pain in endometriosis | Prostaglandin synthesis inhibitionMagnesium plays multiple relevant roles in endometriosis management: it inhibits prostaglandin synthesis (reducing uterine cramping), relaxes smooth muscle (reducing the spasmodic component of pelvic pain), modulates NMDA receptors in the central nervous system (relevant to central sensitisation and chronic pain amplification), and supports progesterone production (relevant to the oestrogen-progesterone imbalance of endometriosis).
A 2017 Cochrane review found moderate-quality evidence that magnesium supplementation significantly reduces primary dysmenorrhoea — the mechanism is directly relevant to endometriosis-associated pain. Magnesium deficiency is widespread in India due to reliance on refined grains (which strip magnesium in processing). Best dietary sources: pumpkin seeds, dark chocolate (70%+), almonds, spinach, methi (fenugreek), whole grains. Supplemental form: magnesium glycinate (most bioavailable and least laxative) 200–400mg nightly.
⚗️ Cochrane 2017: magnesium reduces dysmenorrhoea | NMDA modulation for central sensitisationVitamin D receptors are present on immune cells, endometrial cells, and the endometriotic lesions themselves. Multiple studies have found that women with endometriosis have lower Vitamin D levels than controls, and that Vitamin D exerts direct anti-endometriotic effects through: inhibition of endometrial cell proliferation, reduction of inflammatory cytokine production in lesions, enhancement of NK cell function (the cells that fail to clear ectopic endometrial tissue), and regulation of aromatase expression (relevant to the local oestrogen production of lesions).
A 2016 randomised trial published in the Iranian Journal of Reproductive Medicine found that weekly high-dose Vitamin D supplementation significantly reduced dysmenorrhoea and NSAID use in endometriosis patients over 8 weeks. India has paradoxically high rates of Vitamin D deficiency despite abundant sunshine — due to indoor lifestyles, skin coverage, and darker skin requiring longer sun exposure for equivalent synthesis. Testing and correcting Vitamin D status is a low-risk, high-potential intervention for Indian women with endometriosis.
⚗️ VDR on lesions + NK cells | RCT: Vit D reduces dysmenorrhoea in endometriosis | Aromatase regulationThe gut microbiome plays a direct role in oestrogen metabolism through the estrobolome — the collection of gut bacteria producing enzymes (particularly beta-glucuronidase) that deconjugate oestrogen in the gut, determining whether oestrogen is excreted or reabsorbed into circulation. Dysbiosis of the estrobolome — reduced microbial diversity, overgrowth of high-beta-glucuronidase species — leads to increased oestrogen recirculation, which may fuel endometriosis lesion growth.
Emerging research has found that women with endometriosis have measurably different gut and peritoneal microbiomes compared to controls. Supporting gut microbiome diversity through dietary fibre, fermented foods (homemade curd, chaas, idli/dosa), and reducing antibiotic use may contribute to oestrogen regulation through the gut-endometriosis axis. This is an area of active research — practical steps are low-risk and align with general gut health guidance. Read more: Gut Health and Overall Wellness
⚗️ Estrobolome beta-glucuronidase | Altered microbiome in endometriosis patients | Oestrogen recirculation pathwayRegular moderate exercise benefits endometriosis through multiple pathways: it reduces circulating oestrogen levels (adipose tissue is a major oestrogen production site — exercise that reduces body fat reduces oestrogen burden), it releases endorphins and endocannabinoids that provide natural analgesia, it reduces prostaglandin production, it improves immune function (including NK cell activity), and it reduces systemic inflammation through myokine signalling.
A 2017 systematic review in the Journal of Physical Therapy Science found that regular aerobic exercise significantly reduced dysmenorrhoea severity and duration. Yoga — particularly practices addressing pelvic floor tension and nervous system regulation — has emerging evidence for endometriosis pain management. A 2017 Brazilian RCT found that a specific yoga programme significantly improved quality of life and reduced pain scores in women with endometriosis over 8 weeks.
During acute pain flares: gentle movement (walking, restorative yoga, stretching) is preferable to complete rest — which worsens deconditioning and increases central pain sensitisation. During pain-free periods: build cardiovascular fitness through activities you enjoy — swimming, cycling, yoga, dance, walking. Exercise should feel nourishing, not punishing.
⚗️ Exercise → reduced oestrogen + endorphins + NK cell function | Yoga RCT: pain score reduction (Brazil 2017)Continuous low-level topical heat (a heating pad at 38–40°C applied to the lower abdomen) produces pain relief for dysmenorrhoea comparable to ibuprofen in controlled studies — through cutaneous thermoreceptor activation that closes the pain gate mechanism and directly reducing muscle spasm. The 2001 American Journal of Obstetrics and Gynecology study found continuous heat as effective as ibuprofen for primary dysmenorrhoea — the mechanism applies to endometriosis-associated pelvic pain.
Transcutaneous electrical nerve stimulation (TENS) — particularly high-frequency TENS applied to the lower abdomen or acupuncture points — has Cochrane-reviewed evidence for dysmenorrhoea reduction. It works by activating A-beta sensory nerve fibres that competitively inhibit pain signal transmission through the dorsal horn of the spinal cord (gate control theory). Home TENS units are widely available and carry no pharmacological side effects.
Chronic psychological stress is not merely “something that makes endometriosis feel worse.” It has measurable immunological effects directly relevant to the disease. Cortisol — produced by the HPA axis in response to stress — suppresses NK cell activity (already impaired in endometriosis), increases pro-inflammatory cytokine production, and raises cortisol-driven insulin resistance that may worsen oestrogen dominance. Women with endometriosis have documented HPA axis dysregulation.
Mindfulness-based stress reduction (MBSR), yoga, pranayama, meditation, and adequate sleep are not just wellness recommendations — they are physiologically grounded interventions that reduce HPA axis activation and improve the immune environment relevant to endometriosis. A 2017 study found that an 8-week MBSR programme significantly reduced pain, anxiety, and quality-of-life impairment in women with endometriosis. For Indian women: the rich tradition of pranayama, yoga nidra, and meditation provides culturally resonant tools for this purpose.
⚗️ Cortisol suppresses NK cells | HPA dysregulation in endometriosis | MBSR 8-week trial: pain reductionEndometriosis Myths vs. Facts — The Ones That Delay Diagnosis and Cause Harm
“Painful periods are normal — all women have them.”
Some menstrual discomfort is common. Pain severe enough to miss school, work, or daily activities — or pain that requires strong pain relief and is not fully controlled by it — is not normal. It is a symptom requiring investigation. This cultural normalisation of menstrual pain is the single biggest driver of the 7–10 year diagnostic delay globally.
“Pregnancy cures endometriosis.”
Pregnancy suppresses endometriosis symptoms temporarily (high progesterone levels during pregnancy suppress lesion activity). It does not eradicate the disease. Symptoms return after delivery and the completion of breastfeeding as oestrogen levels recover. Advising a woman to “get pregnant” as a treatment for endometriosis is medically inaccurate and ethically problematic. It also ignores the fertility challenges endometriosis itself may be causing.
“A normal ultrasound means you don’t have endometriosis.”
Transvaginal ultrasound cannot detect peritoneal endometriosis — the most common form — or small lesions. A normal ultrasound does not exclude endometriosis. Only laparoscopy with biopsy provides definitive diagnosis. Many women are falsely reassured by a normal ultrasound and sent home without further investigation despite significant symptoms.
“If you have Stage 1 endometriosis, your pain should be mild.”
Endometriosis staging (I–IV) reflects anatomical disease burden, not pain severity. Pain is driven by the inflammatory environment (prostaglandin and cytokine levels), nerve infiltration by lesions, and central sensitisation — none of which correlate linearly with anatomical stage. Women with Stage 1 disease can be severely disabled; women with Stage 4 disease can have minimal symptoms.
“Hysterectomy will cure endometriosis.”
Hysterectomy removes the uterus — not the peritoneal lesions. If peritoneal or ovarian endometriosis is not also excised at the time of hysterectomy, residual lesions continue to be stimulated (particularly if ovaries are retained) and symptoms can persist or recur. Hysterectomy with ovarian conservation has a 62% symptom recurrence rate in endometriosis if lesions are not adequately excised. Complete excision of all visible disease is as important as hysterectomy itself.
When to Seek Medical Help — And What to Ask For
Periods that significantly disrupt daily life: Missing school, work, or social activities due to period pain — or requiring bed rest — is not normal and warrants investigation.
Pain that does not respond adequately to ibuprofen or naproxen at standard doses: NSAIDs are the first-line treatment for dysmenorrhoea. Pain that breaks through standard doses is a red flag.
Deep pain during sexual intercourse: This specific symptom — distinct from entry discomfort — is strongly associated with endometriosis, particularly uterosacral and rectovaginal involvement.
Bowel or bladder symptoms that follow your menstrual cycle: Cyclical diarrhoea, constipation, bloating, painful urination, or rectal bleeding at menstruation requires endometriosis investigation — not just IBS management.
Difficulty conceiving after 6–12 months of trying: Endometriosis is a common and treatable cause of infertility. Early investigation allows treatment before ovarian reserve is further compromised.
Family history of endometriosis: First-degree family history with any of the above symptoms warrants earlier, lower-threshold investigation.
When you see a doctor: use the phrase “I am concerned about endometriosis” explicitly. Ask for referral to a gynaecologist if your GP cannot address this. Ask specifically about transvaginal ultrasound with endometriosis protocol. Do not accept “your periods are just heavy” as a complete response to debilitating pain.
Frequently Asked Questions About Endometriosis Symptoms and Treatment
The main symptoms are: severely painful periods (dysmenorrhoea) that disrupt daily life and don’t fully respond to painkillers, chronic pelvic pain throughout the cycle, deep pain during or after sexual intercourse (dyspareunia), painful bowel movements or urination at menstruation, heavy or irregular bleeding, cyclical GI symptoms (bloating, nausea, diarrhoea), chronic fatigue, and infertility or subfertility. Critically: pain severity does not correlate with disease stage — severe pain can occur with minimal visible disease.
The global average is 7–10 years from symptom onset. In India, the delay is likely longer due to cultural normalisation of period pain and limited specialist access. Definitive diagnosis requires laparoscopy with biopsy. A normal ultrasound does not exclude endometriosis. Women should explicitly name endometriosis as a concern when seeking medical care and request referral to a gynaecologist experienced with the condition.
Natural approaches are effective adjuncts to medical management. The strongest evidence is for: anti-inflammatory diet (reducing trans fats and red meat, increasing omega-3 and cruciferous vegetables), omega-3 supplementation (meta-analysis: significant pain reduction), magnesium (Cochrane evidence for dysmenorrhoea), Vitamin D correction (RCT: significant dysmenorrhoea reduction), regular exercise (reduces oestrogen and improves immune function), and stress management (MBSR reduces pain and quality-of-life impairment). These are adjuncts — medical diagnosis and treatment should not be delayed or replaced by natural approaches alone.
No. While 30–50% of women with endometriosis experience fertility challenges, many conceive naturally or with assistance. Fertility impact depends on disease location (tubal, ovarian, or causing adhesions has greater impact), stage, age, and treatment. Women with endometriosis who wish to conceive should seek consultation with a reproductive endocrinologist for personalised assessment. Early diagnosis and treatment generally improves fertility outcomes.
PCOS is a hormonal-metabolic disorder driven by insulin resistance, causing irregular ovulation and androgen excess. Endometriosis is an immune-inflammatory oestrogen-driven condition where endometrial-like tissue grows outside the uterus. They can co-exist. Key differences: PCOS typically causes irregular periods; endometriosis causes severe pain with periods. PCOS presents with androgen symptoms (acne, hair growth); endometriosis does not. Both can cause infertility through different mechanisms. Treatment approaches are distinct.
Foods associated with worsening endometriosis: red and processed meat (prostaglandin-promoting + potential dioxin bioaccumulation), trans fats (directly elevate inflammatory markers — strongest dietary evidence), refined sugar (worsens inflammatory markers), excess alcohol (raises circulating oestrogen), and excess caffeine (some studies link to higher oestrogen). Foods to increase: omega-3-rich foods (walnuts, flaxseed, mustard oil), cruciferous vegetables (oestrogen metabolism support), high-fibre foods (oestrogen elimination), and turmeric with black pepper daily.
Related Articles You’ll Love
You have been told, perhaps for years, that your pain is normal. That all women feel this way. That you are being dramatic, or anxious, or sensitive. Endometriosis is none of those things. It is a real, documented, physiologically complex disease that affects 190 million women worldwide — and receives less research funding per patient than almost any condition of comparable prevalence.
Your pain is real. It deserves investigation. And you deserve a diagnosis that names what is happening in your body, not a prescription for ibuprofen and patience.
Trust your body. Advocate for yourself. And share this with every woman you know who has been told her pain is “just periods.” 🌸Which part of the endometriosis science surprised you most — the immune dysfunction mechanism, the 7–10 year diagnostic delay, or the fact that disease stage doesn’t predict pain? Share this guide — it may reach someone who has been waiting years for someone to explain what is happening to them. 👇
Sources & Further Reading
- World Endometriosis Research Foundation — Global Prevalence: 190 Million Women
- Human Reproduction Update (2010) — Retrograde Menstruation and Endometriosis: NK Cell Failure Hypothesis
- Nurses’ Health Study II — Omega-3 and Trans Fat Intake and Endometriosis Risk
- AJOG (2011) — Fish Oil Supplementation and Dysmenorrhoea in Endometriosis: Clinical Trial
- Cochrane Review (2017) — Magnesium for Primary Dysmenorrhoea
- AJOG (2001) — Continuous Heat as Effective as Ibuprofen for Dysmenorrhoea
- Journal of Physical Therapy Science (2017) — Exercise and Dysmenorrhoea: Systematic Review
- Iranian Journal of Reproductive Medicine (2016) — Vitamin D Supplementation and Dysmenorrhoea in Endometriosis: RCT
- HerbeeLife — Health Benefits of Turmeric: The NF-kB Anti-Inflammatory Mechanism
- HerbeeLife — Natural Health & Ayurvedic Wellness
Disclaimer: This content is for informational purposes only and does not constitute medical advice. Endometriosis requires professional diagnosis and individualised medical management. Always consult your gynaecologist or healthcare provider for diagnosis and treatment decisions. Read full disclaimer →



