Hypothyroidism is one of the most prevalent yet most underdiagnosed chronic conditions affecting Indian women. Estimates suggest that approximately 1 in 10 Indian women has some form of thyroid dysfunction — with hypothyroidism being the most common — and that the majority of cases remain either undiagnosed or inadequately managed. The symptoms are insidious and accumulate gradually: the fatigue that is attributed to parenting demands, the weight that is blamed on ageing, the hair fall that is assumed to be stress, the depression that is dismissed as personality, the constipation that is managed with laxatives rather than investigated as a thyroid sign.
Understanding hypothyroidism symptoms and causes at the depth this condition demands — the molecular biology of thyroid hormone production, the immune mechanisms of Hashimoto’s thyroiditis, the nutritional requirements of thyroid function, the difference between TSH-normal and T3-deficient states, and the specific lifestyle and dietary interventions that support thyroid health — is what separates someone who manages their condition effectively from someone who remains symptomatic on medication because the underlying drivers were never addressed.
This guide provides that depth — for the millions of Indian women whose thyroid condition deserves better than a prescription and a six-monthly blood test.
What the Thyroid Does — Why Its Function Affects Everything
The thyroid gland — a butterfly-shaped gland weighing approximately 25 grams, located at the base of the neck — produces the thyroid hormones thyroxine (T4) and triiodothyronine (T3) that regulate the metabolic rate of virtually every cell in the human body. Unlike most hormones that target specific tissues, thyroid hormones have receptors in nearly every organ and cell type — making them the most broadly acting biochemical regulators in human physiology.
The thyroid’s regulatory reach is extraordinary: heart rate and cardiac output, body temperature maintenance, basal metabolic rate, oxygen consumption, protein synthesis, fat metabolism, carbohydrate metabolism, gut motility, skin renewal, hair follicle cycling, brain function (cognition, mood, and neurological development in children), reproductive hormone balance, and bone turnover are all directly governed by thyroid hormone levels. When thyroid hormone production is insufficient — hypothyroidism — every one of these processes is affected to varying degrees, producing the remarkably broad symptom complex that characterises the condition.
The hormone production pathway begins in the hypothalamus (which releases TRH — thyrotropin-releasing hormone in response to falling T3/T4 levels), proceeds to the pituitary (which releases TSH — thyroid-stimulating hormone in response to TRH), and culminates in the thyroid gland itself (which takes up iodine from the bloodstream, combines it with the amino acid tyrosine to form T4 and T3, and secretes them into the circulation). TSH — the pituitary’s signal to the thyroid — is the primary measure used in thyroid function testing: elevated TSH indicates the pituitary is working harder to stimulate a failing thyroid (hypothyroidism), while depressed TSH indicates either excessive thyroid hormone production or exogenous hormone administration.
A critically important and widely misunderstood aspect of thyroid physiology: the thyroid secretes primarily T4 (approximately 80% of output), which is metabolically relatively inactive and serves as a prohormone. T4 must be converted to T3 — the biologically active hormone — in peripheral tissues (primarily the liver, kidney, and muscle) through the enzyme deiodinase. Many people with “normal” TSH and T4 levels have impaired T4-to-T3 conversion and are functionally hypothyroid at the cellular level — experiencing full hypothyroid symptoms with blood tests that appear reassuring. Understanding this conversion step is essential for understanding why some people remain symptomatic on standard levothyroxine (T4) therapy.
Hypothyroidism Symptoms — The Full Picture Beyond Fatigue and Weight Gain
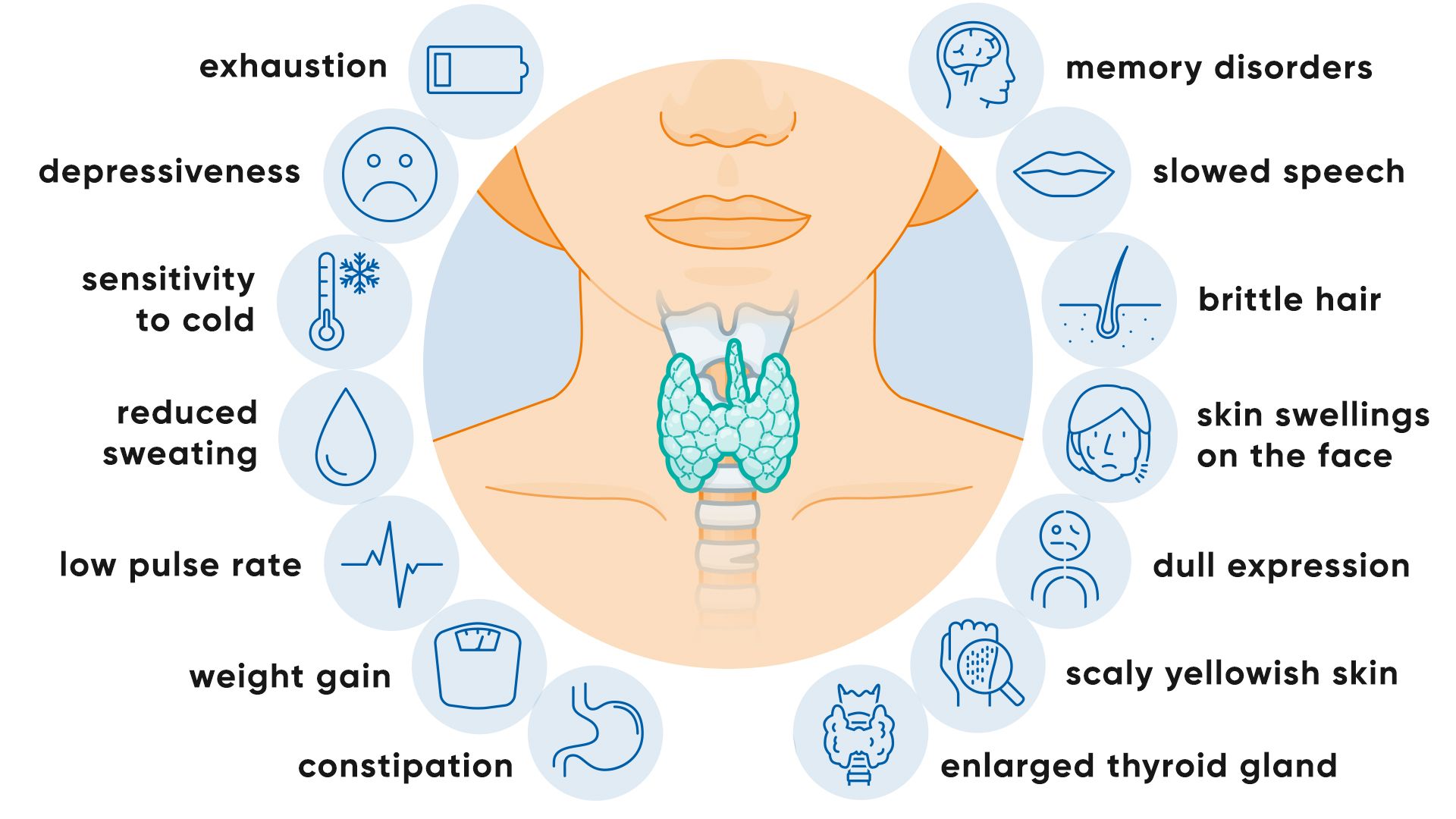
The symptom profile of hypothyroidism reflects the universal metabolic regulatory role of thyroid hormones — virtually every system in the body functions more slowly and less efficiently in a hypothyroid state.
Metabolic and Energy Symptoms
Fatigue and lethargy are the most reported hypothyroidism symptoms — but the character of thyroid fatigue is specific and distinguishable from fatigue of other causes: it is present from the moment of waking (not improved by rest), is accompanied by a general sense of mental and physical heaviness, and does not respond to caffeine with normal alertness-generating effect. The mechanism is direct: thyroid hormones are required for mitochondrial energy production — T3 stimulates the uncoupling protein in mitochondria that governs the efficiency of ATP synthesis. Without adequate T3, mitochondria produce less ATP from the same substrate, reducing the cellular energy currency that drives every metabolic function.
Cold intolerance — feeling cold when others are comfortable, requiring extra layers, having persistently cold hands and feet — results from the reduced thermogenesis (heat production) that accompanies reduced metabolic rate. Thyroid hormones govern basal thermogenesis through their effects on sodium-potassium ATPase activity (the primary heat-generating futile cycle in the body) and through UCP1 (uncoupling protein) activation in brown adipose tissue.
Weight gain — often modest (2–5kg typically) and partially reversible with hormone normalisation — results from reduced basal metabolic rate (the amount of energy expended at rest), reduced thermogenesis, fluid retention, and reduced intestinal motility reducing food transit efficiency. The weight gain of hypothyroidism is characteristically difficult to address through dietary restriction or exercise alone, because the metabolic suppression means fewer calories are needed to maintain body weight — any restriction that would produce weight loss in a euthyroid (normal thyroid) person produces less weight change in a hypothyroid state.
Dermatological and Hair Symptoms
Dry skin results from reduced sebaceous and eccrine gland activity (thyroid hormones regulate the secretory function of skin glands), reduced skin cell turnover (thyroid hormones are required for keratinocyte proliferation), and the fluid shift from intravascular to interstitial compartments (the myxoedema of severe hypothyroidism — non-pitting oedema from glycosaminoglycan accumulation in the dermis — produces a characteristic doughy, pale skin appearance).
Hair loss and texture changes are among the most distressing hypothyroidism symptoms for women — with diffuse hair thinning, loss of the outer third of the eyebrows (a classic clinical sign), and hair that becomes coarse, brittle, and slow-growing. Thyroid hormones are required for the anagen (growth) phase of the hair cycle — hypothyroidism prolongs the telogen (resting/shedding) phase and shortens the anagen phase, producing diffuse telogen effluvium that presents as generalised thinning rather than focal bald patches. The specific connection between thyroid function and hair fall in Indian women is explored in our article on hair fall after 30 in women.
Cardiovascular Symptoms
Slow heart rate (bradycardia) — often noticed as a subjective sense of reduced cardiac effort or palpitations at lower heart rates — results from reduced cardiac contractility and slowed sinoatrial node firing rate. Thyroid hormones directly regulate cardiac β-adrenergic receptor sensitivity, myosin heavy chain isoform expression, and calcium transport in cardiac myocytes.
Elevated cholesterol is one of the most clinically significant cardiovascular features of hypothyroidism — and one that is frequently managed with statins without investigating the thyroid basis. Thyroid hormones are required for the expression of LDL receptors on hepatocytes and for the conversion of cholesterol to bile acids. In hypothyroidism, LDL receptor expression is reduced, cholesterol clearance from the bloodstream is impaired, and both LDL and total cholesterol rise — sometimes dramatically. Treatment of the underlying hypothyroidism often normalises cholesterol levels without statins.
Neurological and Psychological Symptoms
Depression, anxiety, and cognitive impairment (“brain fog”) are among the most debilitating hypothyroidism symptoms and the most frequently misattributed to psychiatric or lifestyle causes. Thyroid hormones are required for serotonin synthesis (T3 stimulates tryptophan hydroxylase, the rate-limiting enzyme in serotonin production), for GABA receptor sensitivity, and for the neuronal myelination, synaptic plasticity, and neurotransmitter recycling that underpin cognitive function. Hypothyroid patients show measurable deficits in memory, processing speed, executive function, and working memory — deficits that partially (and sometimes completely) reverse with adequate thyroid hormone replacement.
Nerve-related symptoms — paraesthesias (tingling, numbness), carpal tunnel syndrome, and slower deep tendon reflexes — result from reduced metabolic activity in peripheral nerve myelin and the deposition of glycosaminoglycans in the carpal tunnel (which compresses the median nerve). These symptoms are frequently investigated and managed as isolated neuropathies without thyroid evaluation — missing the systemic cause.
Reproductive and Hormonal Symptoms in Women
The interaction between thyroid function and reproductive hormones is particularly significant and particularly relevant for women. Thyroid hormones are required for progesterone synthesis, for normal menstrual cycle regulation, and for fertility — hypothyroidism produces menstrual irregularity (both heavy periods and cycle lengthening), impaired ovulation, increased miscarriage risk, and reduced fertility through specific hormonal mechanisms including elevated prolactin (from TRH stimulation of prolactin secretion), which suppresses GnRH and impairs ovulation.
The complete picture of thyroid-reproductive hormone interactions is covered in our article on how hormones affect women’s health — where the HPT (hypothalamic-pituitary-thyroid) axis interaction with the HPG (hypothalamic-pituitary-gonadal) axis and the HPA (stress) axis is explored in depth.
Causes of Hypothyroidism — The Complete Picture
Hashimoto’s Thyroiditis — The Most Important and Most Misunderstood Cause
Hashimoto’s thyroiditis (autoimmune thyroiditis) is the most common cause of hypothyroidism in iodine-sufficient regions — accounting for approximately 90% of hypothyroidism cases in urban India and globally. It is an autoimmune condition in which the immune system produces antibodies against thyroid tissue — primarily against thyroglobulin (TgAb) and thyroid peroxidase (TPOAb) — producing progressive lymphocytic infiltration of the thyroid gland, follicular destruction, and declining hormone production over years to decades.
Understanding Hashimoto’s as an autoimmune condition rather than simply a failing thyroid gland has profound implications for management. The thyroid failure is a consequence of immune dysregulation — the immune system attacking self-tissue. This means that addressing the immune dysregulation (through the same anti-inflammatory dietary and lifestyle interventions that support immune balance in all autoimmune conditions) is the most fundamental treatment available, not merely replacing the hormone that the destroyed thyroid can no longer produce. Natural thyroid support is therefore most impactful when it addresses the autoimmune driver rather than simply supplementing the downstream deficiency.
Hashimoto’s is 7–10 times more common in women than men — reflecting the well-documented sex difference in autoimmune disease susceptibility that oestrogen’s immune-modulating effects partly explain. Its prevalence increases with age, and it frequently coexists with other autoimmune conditions including type 1 diabetes, coeliac disease, rheumatoid arthritis, and vitiligo — a clustering that reflects shared immune dysregulation pathways.
The Hashimoto’s-gut connection is one of the most clinically important insights from modern autoimmunity research: increased intestinal permeability (leaky gut) allows undigested food antigens (particularly gluten from wheat) and bacterial components to enter the systemic circulation, triggering the immune activation that — in genetically predisposed individuals — can target thyroid tissue through molecular mimicry (shared antigenic sequences between foreign proteins and thyroid antigens). This mechanism explains both why gluten sensitivity is significantly more prevalent in Hashimoto’s patients than in the general population, and why gut health improvement produces measurable reductions in thyroid antibody levels in some patients. The gut-thyroid axis connects to the framework in our guide to improving digestion naturally.
Iodine Deficiency and Iodine Excess
Iodine is the essential micronutrient from which thyroid hormones (T4 contains 4 iodine atoms, T3 contains 3) are synthesised — and both deficiency and excess can cause hypothyroidism through different mechanisms.
Iodine deficiency — historically the world’s most common cause of hypothyroidism and goitre — has been substantially reduced in India through the iodised salt programme, but remains relevant in mountain-belt communities (Himachal Pradesh, Uttarakhand, parts of the Northeast) with iodine-depleted soil and water, in populations using non-iodised rock salt (sendha namak), and in people consuming high amounts of goitrogenic foods that block iodine utilisation without adequate iodine intake.
Iodine excess — less intuitive but equally important — can paradoxically induce or worsen hypothyroidism, particularly in individuals with underlying Hashimoto’s. The Wolff-Chaikoff effect describes the transient inhibition of thyroid hormone synthesis that occurs with acute iodine excess — normally a protective transient mechanism, but in individuals with autoimmune thyroid disease, this inhibition can become persistent. This is the reason that excessive iodine supplementation (through high-dose iodine supplements or seaweed products like kelp, which contain extremely high iodine concentrations) is potentially harmful rather than helpful for Hashimoto’s patients.
Nutritional Deficiencies Beyond Iodine
Several nutritional deficiencies beyond iodine are critically important causes and contributors to hypothyroidism that the original article entirely omits — and that represent the most actionable natural interventions for thyroid health.
Selenium is required for the deiodinase enzymes that convert T4 to the active T3 — making selenium the most important micronutrient for the T4-to-T3 conversion that determines how much functional thyroid hormone actually reaches cells. Selenium is also required for the selenoprotein glutathione peroxidase that protects thyroid follicular cells from the hydrogen peroxide generated during thyroid hormone synthesis — selenodeficient thyroid cells are more susceptible to the oxidative damage that drives Hashimoto’s inflammation. Clinical studies have found that selenium supplementation (200mcg daily) significantly reduces TPOAb levels in Hashimoto’s patients, suggesting genuine immunomodulatory effects on the autoimmune process itself. The selenium-selenium food connection to the anti-inflammatory nutritional support is in our anti-inflammatory foods guide.
Iron deficiency impairs thyroid peroxidase (TPO) enzyme activity — TPO is an iron-dependent heme enzyme required for iodine oxidation in thyroid hormone synthesis. Iron deficiency therefore directly impairs thyroid hormone production through its effects on the same enzyme that Hashimoto’s antibodies target. The concurrent iron deficiency and thyroid dysfunction in many Indian women creates a bidirectional impairment: thyroid dysfunction reduces iron absorption (hypothyroidism reduces gastric acid production needed for non-haem iron absorption), and iron deficiency impairs thyroid hormone synthesis.
Vitamin D deficiency — as covered in our immune system guide, affecting 50–94% of urban Indians — has a specific role in thyroid autoimmunity: Vitamin D receptors are expressed on thyroid cells and immune cells, and Vitamin D has immunomodulatory effects that reduce the Th1-mediated autoimmune inflammation of Hashimoto’s. Research shows significant inverse correlations between serum Vitamin D levels and TPOAb levels in Hashimoto’s patients — the lower the Vitamin D, the higher the antibodies.
Zinc is required for the conversion of T4 to T3 in peripheral tissues and for thyroid hormone receptor binding in target cells. Zinc deficiency therefore impairs both the production of active T3 and the cellular response to whatever T3 is produced — a double impairment with functional hypothyroid consequences even when blood tests appear normal.
Stress and the HPA-HPT Axis Interaction
Chronic psychological stress produces measurable suppression of thyroid function through multiple mechanisms — the HPA-HPT axis crosstalk that produces what is sometimes called “functional hypothyroidism” in the context of chronic stress. Cortisol (chronically elevated in the stress state) directly inhibits TSH secretion from the pituitary, reduces T4-to-T3 conversion in peripheral tissues (cortisol activates reverse T3 production instead, producing a metabolically inactive T3 competitor that occupies T3 receptors without activating them), and increases T3 degradation. The result is lower functional T3 activity at cellular receptors despite potentially normal TSH and T4 blood levels.
This stress-thyroid interaction explains the well-documented observation that thyroid symptoms frequently worsen during periods of intense psychological stress even in people on stable levothyroxine doses — the problem is not reduced T4 delivery (which levothyroxine provides) but impaired T4-to-T3 conversion in the liver and periphery due to cortisol excess. Addressing stress through the holistic framework is therefore a direct thyroid health intervention, not merely a supportive lifestyle recommendation. The complete stress-cortisol-thyroid interaction is covered in our article on how hormones affect women’s health.
Subclinical Hypothyroidism — The Missed Diagnosis Affecting Millions
Subclinical hypothyroidism (SCH) is defined by elevated TSH (above the normal range, typically 4.5 mIU/L) with normal T4 levels — indicating that the pituitary is working harder to stimulate the thyroid to maintain normal T4 output, but the compensatory effort is insufficient. Many people with SCH have significant hypothyroid symptoms — the clinical manifestation of a thyroid gland that is failing to maintain normal function despite maximal pituitary stimulation.
SCH is significantly more prevalent in Indian women than previously appreciated, with population studies in India finding rates of 4–10% — and much higher rates in women of reproductive age and postpartum. The decision about whether to treat SCH with levothyroxine is nuanced and should involve an endocrinologist, but the natural thyroid support strategies in this guide are appropriate and potentially beneficial for SCH regardless of the treatment decision.
Additionally, many practitioners and researchers argue that the conventional TSH “normal range” (typically 0.5–4.5 mIU/L) is too broad — and that an optimal TSH for metabolic function and symptom freedom is closer to 1.0–2.5 mIU/L. A patient with a TSH of 4.0 mIU/L is technically “normal” by conventional criteria but may have significant hypothyroid symptoms and suboptimal thyroid function by functional standards — and may benefit from both natural thyroid support and reassessment of their treatment target with their physician.
Natural Support for Thyroid Health — Nutrition, Lifestyle and Ayurveda
The Thyroid-Supportive Diet
Diet for thyroid health is more nuanced than the simple “eat these, avoid those” format most thyroid guides provide. The most important principles are:
Selenium-rich foods daily: Brazil nuts (one nut provides 70–90mcg — the full daily requirement), sunflower seeds, fish (particularly tuna, sardines, and salmon), eggs, and mushrooms are the best dietary selenium sources. Including these consistently is the single most important dietary intervention for T4-to-T3 conversion and for reducing Hashimoto’s antibody levels through the glutathione peroxidase mechanism.
Adequate but not excessive iodine: Iodised salt (used in cooking in reasonable amounts) provides adequate iodine for most Indians without excess. Seafood provides natural iodine alongside selenium (the natural pairing in ocean environments that supports thyroid function optimally). Avoid high-dose iodine supplements and high-dose seaweed products (kelp) if you have Hashimoto’s — excess iodine can worsen the autoimmune process.
Iron-rich foods with absorption enhancers: The iron-thyroid connection makes iron optimisation a direct thyroid health intervention. Non-haem iron from legumes and dark leafy greens is significantly better absorbed when consumed with Vitamin C (amla, lemon, fresh peppers). Our blood sugar and anti-inflammatory nutrition guides cover food sequencing that maximises non-haem iron absorption.
The goitrogen myth and reality: Cruciferous vegetables (broccoli, cauliflower, cabbage, radish, mustard greens) contain glucosinolates that are converted to compounds that can mildly inhibit thyroid hormone synthesis when consumed raw in very large quantities in iodine-deficient individuals. For most people with adequate iodine intake, cruciferous vegetables pose no meaningful thyroid risk — the anti-inflammatory, sulforaphane-containing benefits of cruciferous vegetables (covered in our liver cleanse guide) are far more relevant to thyroid health than the theoretical goitrogenic concern. Cooking cruciferous vegetables largely eliminates goitrogenic compounds. Completely avoiding these highly beneficial vegetables based on thyroid concerns is not warranted for most people.
Anti-inflammatory whole food pattern: The autoimmune basis of most hypothyroidism means anti-inflammatory nutrition is a direct therapeutic intervention. The dietary framework in our anti-inflammatory foods guide — diverse plant foods, omega-3-rich fish, turmeric, ginger, olive oil, minimal refined carbohydrates and processed foods — directly reduces the inflammatory cytokine environment that drives Hashimoto’s autoimmune activity. The connection between gut health, leaky gut, and Hashimoto’s specifically makes the probiotic and prebiotic strategies in our digestion guide directly relevant to thyroid autoimmune management.
Ayurvedic Thyroid Support — The Kaphaja Galaganda Framework
Ayurveda classifies thyroid conditions (particularly goitre and related thyroid dysfunction) as Galaganda — a swelling in the neck region — with hypothyroidism most closely corresponding to a Kapha-Vata aggravation pattern characterised by the heaviness, coldness, slowness, and congestion that reflect both Kapha excess and the Vata-driven irregularity of thyroid hormone secretion.
The Ayurvedic treatment approach for thyroid dysfunction focuses on three interconnected strategies: Kapha and Vata reduction through warming, light, stimulating herbs and foods; restoration of metabolic fire (Agni) through digestive support that both addresses the gut-thyroid axis and directly improves cellular metabolism; and Rasayana (rejuvenative) herbs that support the neuroendocrine axis governing thyroid function.
Ashwagandha (Withania somnifera) is the most important Ayurvedic adaptogen for thyroid health — and has the strongest research evidence of any herb for directly supporting thyroid function. A randomised double-blind placebo-controlled trial published in the Journal of Alternative and Complementary Medicine found that ashwagandha root extract (600mg daily) significantly increased serum T3 and T4 levels and significantly reduced TSH in subclinical hypothyroid patients over 8 weeks — representing genuine evidence that ashwagandha can directly improve thyroid hormone levels, not just ameliorate symptoms. The proposed mechanisms include modulation of the HPT axis through withanolide effects on the pituitary, reduction of cortisol-driven T4-to-T3 conversion impairment, and direct stimulation of thyroid hormone synthesis. The comprehensive evidence for ashwagandha is covered in our article on ashwagandha for stress and anxiety — the same stress-reducing and adaptogenic effects are the primary mechanism for thyroid support.
Guggul (Commiphora mukul) — a resin from the guggul tree used in classical Ayurveda for thyroid and metabolic conditions — contains guggulsterones that have documented effects on thyroid hormone metabolism, stimulating iodine uptake and increasing T3 levels in research settings. A study published in the Journal of Ethnopharmacology found guggul extract significantly increased T3 levels in animal models of hypothyroidism. While human clinical trials are limited, the traditional classification of guggul as a Kaphahara (Kapha-reducing) and Agni-stimulating herb directly corresponds to its proposed thyroid-activating mechanism.
Gokshura (Tribulus terrestris) is used in Ayurvedic practice to support the reproductive-thyroid hormone axis — relevant for women whose hypothyroidism is contributing to fertility or menstrual cycle disruption. The steroidal saponins in gokshura support DHEA and testosterone precursor availability, which through the hormone-conversion pathway supports the oestrogen-progesterone balance that is disrupted by hypothyroidism.
The Role of Exercise in Thyroid Support
Regular moderate exercise is one of the most important lifestyle interventions for thyroid health — through multiple complementary mechanisms. Exercise increases cellular T3 receptor sensitivity, improving the cellular response to available T3. It increases AMPK activity (the cellular energy sensor) which supports thyroid hormone signalling efficiency. It reduces cortisol chronically (improving T4-to-T3 conversion). And the cardiovascular, metabolic, and mood benefits of regular exercise directly address the symptoms of hypothyroidism — fatigue, weight gain, depression, and cardiovascular risk — through pathways that complement thyroid hormone replacement rather than depending on it.
The comprehensive exercise evidence is in our article on benefits of regular exercise for longevity and wellbeing. For hypothyroid patients specifically, building exercise capacity gradually is important — the fatigue and muscle weakness of hypothyroidism make sudden vigorous exercise poorly tolerated and potentially counterproductive if it significantly elevates cortisol without the cardiovascular fitness to manage the response. Starting with yoga and walking, building to resistance training and moderate aerobic exercise over months, is the most appropriate progression.
Sleep, Circadian Rhythm, and Thyroid Health
TSH secretion follows a strong circadian pattern — peaking between 11pm and 4am during sleep and reaching its nadir in the afternoon. This nocturnal TSH surge is the primary stimulus for thyroid hormone synthesis replenishment, meaning that consistently poor or insufficient sleep directly impairs the TSH-driven thyroid stimulation that maintains hormone levels through the day. Chronic sleep deprivation is associated with measurably reduced T3 levels and impaired thyroid hormone production in multiple research studies. Maintaining consistent, adequate sleep — the most frequently prescribed and least frequently implemented thyroid health recommendation — is not merely supportive care but a direct thyroid function intervention. The comprehensive sleep and circadian health framework is in our healthy morning routine guide.
Understanding Your Thyroid Tests — Beyond TSH
Many people with thyroid conditions are managed on TSH alone — a significant limitation given that TSH reflects pituitary function and does not directly measure the T3 that actually drives cellular metabolic effects. A comprehensive thyroid assessment should include:
TSH — the standard screening and monitoring test. Optimal range (for symptomatic assessment, not merely “within normal”): 1.0–2.5 mIU/L. Many patients feel significantly better when TSH is in the lower half of the normal range rather than the upper half.
Free T4 (FT4) — the unbound, bioavailable thyroxine that is converted to T3 in tissues. Low normal FT4 with high TSH confirms primary hypothyroidism. Normal FT4 with symptoms suggests conversion impairment.
Free T3 (FT3) — the biologically active hormone. Significantly low FT3 with normal FT4 indicates T4-to-T3 conversion impairment — the most common cause of persistent symptoms on levothyroxine therapy. This test is the most likely to reveal why someone remains symptomatic despite “normal” TSH.
TPOAb and TgAb (thyroid peroxidase antibodies and thyroglobulin antibodies) — elevated levels confirm Hashimoto’s autoimmune thyroiditis as the cause. These tests determine whether the condition requires an autoimmune management approach (addressing the immune dysregulation) rather than simply hormone replacement.
Reverse T3 (rT3) — the inactive isomer of T3 produced when cortisol inhibits normal T4-to-T3 conversion. Elevated rT3 (particularly a low FT3:rT3 ratio) is the biochemical marker of stress-induced thyroid hormone impairment — the “functional hypothyroidism” of chronic stress states that appears on standard tests as normal TSH and T4.
Hypothyroidism: Myth vs. Fact
| ❌ The Myth | ✅ The Truth |
|---|---|
| Once on thyroid medication, you must take it forever with no natural interventions possible | Levothyroxine replaces the hormone a failing thyroid cannot produce — it does not address the autoimmune process destroying the thyroid. Natural interventions (selenium for TPOAb reduction, gut health for leaky gut-triggered autoimmunity, Vitamin D, stress management, anti-inflammatory diet) can significantly slow Hashimoto’s progression, reduce antibody levels, and occasionally reduce medication requirements over time under medical supervision. In early-stage subclinical hypothyroidism, comprehensive natural interventions may delay or prevent the need for pharmaceutical thyroid hormone indefinitely. |
| You must avoid all cruciferous vegetables with hypothyroidism | Cruciferous vegetables are only problematic in raw, very large quantities in iodine-deficient individuals. For most people with adequate iodine intake, cooked cruciferous vegetables pose no meaningful thyroid risk. Their anti-inflammatory sulforaphane compounds, liver-supportive glucosinolates, and gut microbiome-supportive fibre are far more relevant to Hashimoto’s management than their theoretical goitrogenic effect. Complete avoidance of these highly beneficial foods based on a mild concern that does not apply to most patients is unnecessarily restrictive and potentially counterproductive. |
| Normal TSH means your thyroid is fine | TSH measures pituitary signalling to the thyroid — not the active T3 that drives cellular metabolism. A person with normal TSH can have impaired T4-to-T3 conversion (producing low cellular T3 and hypothyroid symptoms), elevated reverse T3 (blocking T3 receptors), positive TPOAb (active Hashimoto’s requiring management), or subclinical T4 deficiency approaching the lower end of normal. Persistent hypothyroid symptoms despite normal TSH warrant further investigation with FT3, FT4, and thyroid antibodies — not dismissal. |
| Thyroid issues are just about the thyroid | Hypothyroidism — particularly Hashimoto’s — is a systemic condition with roots in immune dysregulation, gut health, nutritional status, stress physiology, and environmental factors. Managing it effectively requires addressing all of these contributing systems, not merely replacing the hormone output of the damaged gland. The most successful long-term outcomes in thyroid management integrate pharmaceutical hormone replacement (where indicated) with anti-inflammatory nutrition, gut health optimisation, nutritional deficiency correction, stress management, and regular exercise. |
| Weight gain from hypothyroidism disappears immediately when treatment starts | The metabolic changes of hypothyroidism reverse gradually over weeks to months of optimised thyroid hormone replacement — and the weight gain, which partly reflects genuine adipose accumulation and partly reflects fluid retention, responds differently to treatment. Weight loss on thyroid replacement is usually modest (2–5kg) and may not fully materialise until hormone levels are optimally adjusted rather than merely “normal.” Many patients find that the weight gained with hypothyroidism does not fully normalise until TSH is at the lower end of the normal range and FT3 is optimised. |
| Hypothyroidism is not serious if you take your medication | Undertreated hypothyroidism (persistent symptoms, elevated TSH, suboptimal FT3) is associated with increased cardiovascular risk (elevated LDL, impaired cardiac function), reduced cognitive function, impaired fertility, increased miscarriage risk, depression, and in pregnancy — potentially lifelong neurological developmental consequences for the child. Optimal thyroid management — not just medication compliance but optimised hormone levels, addressed nutritional deficiencies, and reduced autoimmune activity — produces meaningfully better long-term health outcomes than minimum-adequate treatment. |
Hypothyroidism and Pregnancy — The Most Critical Context
Hypothyroidism during pregnancy is one of the most consequential scenarios in thyroid medicine — and one that demands specific, urgent attention beyond the scope of general thyroid management. Thyroid hormones are critical for foetal neurological development, particularly during the first trimester when the foetal thyroid is not yet functional and the foetus depends entirely on maternal T4 for brain development. Untreated or undertreated maternal hypothyroidism during the first trimester is associated with reduced IQ, impaired cognitive development, and neurological abnormalities in the child.
All women with known hypothyroidism should have their levothyroxine dose reviewed at the first antenatal appointment — pregnancy increases thyroid hormone requirements by approximately 30–50% from the first trimester. All women should be screened for thyroid dysfunction when planning pregnancy or at the first antenatal visit. TPOAb-positive women (even with normal TSH) have significantly higher risk of miscarriage and postpartum thyroiditis and require monitoring throughout pregnancy. These are medical priorities that require endocrinologist or obstetrician involvement alongside any natural thyroid support measures.
Related Articles From HerbeeLife
📖 How Hormones Affect Your Health: The Complete Guide Women Must Read
📖 Ashwagandha for Stress and Anxiety: What Actually Happens in Your Body
📖 Hair Fall After 30 Women: 9 Alarming Causes You Must Stop Ignoring
📖 Anti-Inflammatory Foods: 12 Powerful Choices That Fight Disease From the Inside Out
📖 How to Improve Digestion Naturally: 12 Powerful Science-Backed Strategies
📖 Natural Ways to Boost Your Immune System: 12 Science-Backed Strategies
📖 Healthy Morning Routine: 10 Powerful Habits That Transform Your Entire Day
Frequently Asked Questions: Hypothyroidism Symptoms and Causes
Can hypothyroidism be managed naturally without medication?
In subclinical hypothyroidism (mildly elevated TSH with normal T4 and relatively mild symptoms), comprehensive natural interventions — correcting selenium, Vitamin D, iron, and zinc deficiencies; ashwagandha for HPT axis support; anti-inflammatory diet; stress management; gut health optimisation — can normalise TSH and resolve symptoms in some patients, delaying or avoiding pharmaceutical treatment. In overt hypothyroidism (elevated TSH with low T4 and significant symptoms), levothyroxine replacement is generally necessary and appropriate — but natural interventions remain important for addressing the autoimmune basis of Hashimoto’s, reducing antibody levels, and improving the T4-to-T3 conversion that determines how effectively levothyroxine actually works. Natural management complements rather than replaces pharmaceutical treatment in most overt hypothyroid cases.
Why do I still have hypothyroid symptoms on levothyroxine with normal TSH?
The most common reason is impaired T4-to-T3 conversion — levothyroxine provides T4, which must be converted to the active T3 in peripheral tissues. In many patients, this conversion is impaired by selenium deficiency, elevated cortisol (from chronic stress), insulin resistance, liver dysfunction, or nutritional deficiencies — meaning that despite normal TSH (which reflects T4 levels), cellular T3 is insufficient. Getting FT3 and rT3 tested alongside TSH reveals whether conversion impairment is the issue. Addressing the impairment through selenium supplementation, stress management, and liver support (our liver cleanse guide) often resolves persistent symptoms.
Is Hashimoto’s the same as hypothyroidism?
Hashimoto’s thyroiditis is the most common cause of hypothyroidism — but they are distinct conditions. Hashimoto’s is an autoimmune condition (defined by thyroid antibodies) that may or may not have caused hypothyroidism at the time of diagnosis. Early Hashimoto’s with normal thyroid function (TPOAb positive, normal TSH) does not require levothyroxine but benefits enormously from the autoimmune management strategies in this guide. Advanced Hashimoto’s where thyroid destruction has produced hypothyroidism requires both autoimmune management and levothyroxine replacement.
What are the best foods for thyroid health in an Indian diet?
Brazil nuts (selenium — one daily is sufficient), eggs (selenium, iodine, zinc), fish and seafood (iodine, selenium, omega-3), dark leafy greens like moringa and methi (iron, magnesium), legumes (zinc, iron, fibre for gut health), iodised salt in cooking, pumpkin seeds (zinc, selenium), turmeric with black pepper and ghee (anti-inflammatory for Hashimoto’s), and fresh amla (Vitamin C for iron absorption, antioxidant protection). The Indian traditional diet prepared with whole ingredients and diverse spices supports thyroid health well — the challenges arise with the progressive substitution of whole foods with refined and processed alternatives.
Can stress cause hypothyroidism?
Chronic stress does not directly cause structural hypothyroidism (it cannot trigger Hashimoto’s by itself) — but it can cause functional hypothyroidism through two mechanisms: chronically elevated cortisol inhibits T4-to-T3 conversion (producing low cellular T3 despite normal blood T4), and drives rT3 production that blocks T3 receptors. Additionally, the immune dysregulation of chronic stress (particularly the Th1/Th2 imbalance that chronic HPA activation produces) is a known trigger for autoimmune thyroid disease in genetically predisposed individuals. Managing stress is therefore both preventive (reducing autoimmune trigger exposure) and therapeutic (improving T4-to-T3 conversion in existing hypothyroid patients).
Sources and References
1. Unnikrishnan AG, Menon UV. Thyroid disorders in India: An epidemiological perspective. Indian Journal of Endocrinology and Metabolism, 2011.
2. Garber JR et al. Clinical practice guidelines for hypothyroidism in adults. Thyroid, 2012.
3. Toulis KA et al. Selenium supplementation in the treatment of Hashimoto’s thyroiditis: a systematic review and a meta-analysis. Thyroid, 2010.
4. Sharma AK et al. Efficacy and safety of ashwagandha root extract in subclinical hypothyroid patients: a double-blind, randomized placebo-controlled trial. Journal of Alternative and Complementary Medicine, 2018.
5. Mazokopakis EE, Papadomanolaki MG. Effects of 12 months treatment with L-selenomethionine on serum anti-TPO levels in patients with Hashimoto’s thyroiditis. Thyroid, 2007.
6. Kim D. The role of vitamin D in thyroid diseases. International Journal of Molecular Sciences, 2017.
7. Brent GA. Mechanisms of thyroid hormone action. Journal of Clinical Investigation, 2012.
Follow HerbeeLife
🌿 Stay connected for more evidence-based women’s health, thyroid science, and Ayurvedic wellness content:
📸 Instagram | 📌 Pinterest | 👥 Facebook | 🐦 X (Twitter)
Final Thoughts: Your Thyroid Is Not Failing in Isolation — It Is Responding to Its Environment
The thyroid gland does not fail randomly or inevitably. In most cases — particularly in the autoimmune Hashimoto’s that accounts for 90% of hypothyroidism in iodine-sufficient India — it fails in response to a specific set of conditions: immune dysregulation driven by gut permeability and inflammatory diet, nutritional deficiencies in selenium and Vitamin D that deprive thyroid cells of their protective antioxidant enzymes, chronic stress that suppresses T4-to-T3 conversion and drives autoimmune triggers, and the cumulative exposure to environmental thyroid disruptors in modern urban life.
Addressing these conditions — alongside appropriate pharmaceutical management when hormone replacement is genuinely needed — is the most complete and most respectful approach to thyroid health. Not accepting “your thyroid is failing, take this pill forever” as the final answer. But also not abandoning pharmaceutical treatment that provides genuine benefit in the hope that natural interventions alone will be sufficient when the gland is significantly compromised.
The most effective thyroid management is both: precise pharmaceutical support for what the thyroid can no longer produce, combined with the nutritional, lifestyle, and herbal strategies that address why it stopped producing in the first place and give the remaining thyroid tissue every possible advantage.
⚠️ Medical Disclaimer: This article is for informational and educational purposes only and does not constitute medical advice. Hypothyroidism requires diagnosis and management by a qualified endocrinologist or physician. Do not adjust or discontinue thyroid medication without medical supervision. Natural strategies in this guide complement but do not replace professional medical management. Read full disclaimer →
💬 Are you managing hypothyroidism or Hashimoto’s — and has any of the information in this guide (about selenium, T3 conversion, gut health, or ashwagandha) changed how you think about your condition? Share your experience in the comments. The community of women managing thyroid conditions in India deserves a place to exchange real information, not just platitudes.